Is Matter around Us Pure - Revision Notes
CBSE Class 9 Science
Revision Notes
CHAPTER – 2
“Is Matter Around Us Pure”
1. Pure Substance & mixture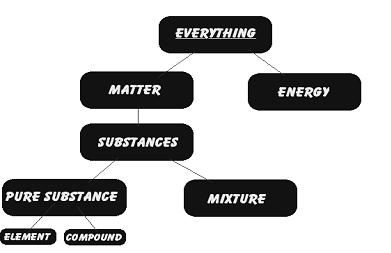
| PURE SUBSTANCE | MIXTURE |
|
|
|
|
|
|
Elements are made up of one kind of atoms only. Compounds are made up of one kind of molecules.
2. Types of Mixtures
Mixtures can also be grouped on the basis of their physical states:
| SOLID | LIQUID | GAS |
SOLID | Salt and sugar | Salt and water | Dust in air |
LIQUID | Mercury and copper | Alcohol and water | Clouds |
GAS | Hydrogen and palladium | Oxygen and water | Air |
ii) on the basis of miscibility:
Homogeneous Mixture | Heterogeneous Mixture |
|
|
|
|
|
|
4. Separating the components of a mixture
The components of a heterogeneous mixture can be separated by
- Simple methods like -hand picking , sieving , & Winnowing
- Special techniques like –
- Evaporation : a mixture of salt and water or sugar and water.
- Centrifugation : Butter from curd, Fine mud particles suspended in water.
- Decantation (Using separating funnel) : Oil from water.
- Sublimation : Camphor from salt,
- Chromatography : Different pigments from an extract of flower petals.
- Distillation and fractional distillation : Separating components of Petroleum
- Magnetic separation: Iron pins from sand.
5. Concentration of Solution
The amount of solute present in a given amount (mass or volume) of solution.
Concentration of a solution = OR
The concentration of a solution can be expressed as mass by mass percentage or as mass by volume percentage.
Mass by mass percentage of a solution =
Mass by volume percentage of a solution =
Types of Solutions
a) on the basis of size of solute particles
True solution | Sol[Colloid] | Suspension |
Homogeneous | Heterogeneous | Heterogeneous |
Size of solute particles is less than 1 n m or | Size of solute particles is between 1 n m or | Size of solute particles is more than |
Particles cannot pass through filter paper. | Particles cannot pass through filter paper. | Particles cannot pass through filter paper. |
Stable | Stable and settle only on centrifugation | Unstable and settle down on its own. |
Solution of sodium chloride in water, suger & water | Milk , Fog | Muddy water, chalk & Water, Smoke in the air. |
Colloidal solution is a heterogeneous mixture. It consists of two phases:-
(i) Dispersed phase : component present in small proportion
(ii) Dispersion medium : component present in large proportion
The particles of colloid are large enough to scatter a beam of light passing through it and make its path visible. Thus, they show Tyndall effect.
The colloidal particles are moving at random in a zigzag motion in all directions.
This type of zig-zag motion of colloidal particles is called Brownian movement.
b) on the basis of amount of solute:
Unsaturated solution -- A solution which has lesser amount of solute that it can dissolve at a given temperature is known as unsaturated solution.
Saturated Solution A solution which has maximum amount of solute that it can dissolve at a given temperature is known as saturated solution
Supersaturated solution A solution which can dissolve amount of solute by increasing temperature saturated solution is known as supersaturated solution.
c) on the basis of nature of solvent
Aqueous solution The solution in which the solvent is water is known as aqueous solution
Non-Aqueous solution The solution in which the solvent is other than water (ether, alcohol or aceton) known as non-aqueous solution.
6. Physical & Chemical Changes
Physical changes - Changes that do not result in the production of a new substance.
- If you melt a block of ice, you still have at the end of the change.
- If you break a bottle, you still have glass.
Examples : melting, freezing, condensing, breaking, crushing, cutting, and bending.
Chemical changes - Changes that result in the production of another substance.
- As in the case of autumn leaves, a change in color is a clue to indicate a chemical change.
- a half eaten apple that turns brown.
7. Alloys
A material that has metallic properties and is composed of two or more chemical elements of which at least one is a metal .
- These cannot be separated into their components by physical methods.
- However, these are considered as mixture because these show the properties of its constituents and can have variable composition.
The benefit of alloys is that you can combine metals that have varying characteristics to create an end product that is stronger, more flexible, or otherwise desirable to manufacturers.
- Aluminium alloys are extensively used in the production of automotive engine parts.
- Copper alloys have excellent electrical and thermal performance, good corrosion resistance, high ductility and relatively low cost.
- Stainless steel alloys are used for many commercial applications such as watch straps, cutlery etc.
- Titanium alloys have high strength, toughness and stiffness & are used in aerospace structures .
You are expected to know
- Types of mixtures.
- Method of Separation of mixtures.
- Types of solutions.
- Concentration terms of solution.
- Physical and Chemical Change.
- Significance of alloys.
