Some Basic Concepts of Chemistry - Solutions
CBSE Class 11 Chemistry
NCERT Solutions
Chapter 1
SOME BASIC CONCEPTS OF CHEMISTRY
1. Calculate the molecular mass of the following:
(i) 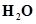
(ii) 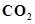
(iii) 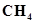
Ans. (i) 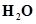 :
:
The molecular mass of water, 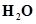
= (2 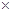 Atomic mass of hydrogen) + (1
Atomic mass of hydrogen) + (1 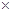 Atomic mass of oxygen)
Atomic mass of oxygen)
= [2(1.0084) + 1(16.00 u)]
= 2.016 u + 16.00 u
= 18.016
= 18.02 u
(ii)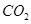 :
:
The molecular mass of carbon dioxide, 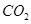
= (1 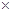 Atomic mass of carbon) + (2
Atomic mass of carbon) + (2 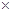 Atomic mass of oxygen)
Atomic mass of oxygen)
= [1(12.011 u) + 2 (16.00 u)]
= 12.011 u + 32.00 u
= 44.01 u
(iii) 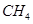 :
:
The molecular mass of methane, 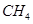
= (1 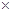 Atomic mass of carbon) + (4
Atomic mass of carbon) + (4 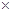 Atomic mass of hydrogen)
Atomic mass of hydrogen)
= [1(12.011 u) + 4 (1.008 u)]
= 12.011 u + 4.032 u
= 16.043 u
2. Calculate the mass percent of different elements present in sodium sulphate (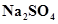 ).
).
Ans. The molecular formula of sodium sulphate is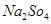 .
.
Molar mass of 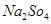 = [(2 × 23.0) + (32.066) + 4 (16.00)]
= [(2 × 23.0) + (32.066) + 4 (16.00)]
= 142.066 g
Mass percent of an element 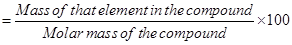
∴ Mass percent of sodium:
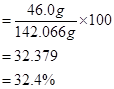
Mass percent of sulphur:
= 22.57
= 23%
Mass percent of oxygen:
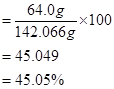
3. Determine the empirical formula of an oxide of iron which has 69.9% iron and 30.1% dioxygen by mass.
Ans. % of iron by mass = 69.9 % [Given]
% of oxygen by mass = 30.1 % [Given]
Relative moles of iron in iron oxide: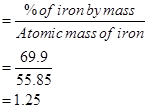
Relative moles of oxygen in iron oxide: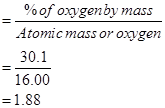
Simplest molar ratio of iron to oxygen:
= 1.25: 1.88
= 1: 1.5
=2: 3
∴ The empirical formula of the iron oxide is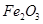 .
.
4. Calculate the amount of carbon dioxide that could be produced when
(i) 1 mole of carbon is burnt in air.
(ii) 1 mole of carbon is burnt in 16 g of dioxygen.
(iii) 2 moles of carbon are burnt in 16 g of dioxygen.
Ans. The balanced reaction of combustion of carbon can be written as:
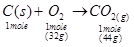
(i) As per the balanced equation, 1 mole of carbon burns in1 mole of dioxygen (air) to produce1 mole of carbon dioxide.
(ii) According to the question, only 16 g of dioxygen is available. Hence, it will react with 0.5 mole of carbon to give 22 g of carbon dioxide. Hence, it is a limiting reactant.
(iii) According to the question, only 16 g of dioxygen is available. It is a limiting reactant. Thus, 16 g of dioxygen can combine with only 0.5 mole of carbon to give 22 g of carbon dioxide.
5. Calculate the mass of sodium acetate 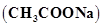 required to make 500 mL of 0.375 molar aqueous solution. Molar mass of sodium acetate is 82.0245 g
required to make 500 mL of 0.375 molar aqueous solution. Molar mass of sodium acetate is 82.0245 g 
Ans. 0.375 M aqueous solution of sodium acetate
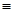 1000 mL of solution containing 0.375 moles of sodium acetate
1000 mL of solution containing 0.375 moles of sodium acetate
∴Number of moles of sodium acetate in 500 mL
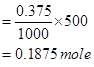
Given
Molar mass of sodium acetate = 82.0245 g 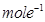
∴ Required mass of sodium acetate = (82.0245 g 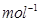 ) * (0.1875 mole)
) * (0.1875 mole)
= 15.38 g
6. Calculate the concentration of nitric acid in moles per litre in a sample which has a density, 1.41 g 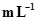 and the mass per cent of nitric acid in it being 69%.
and the mass per cent of nitric acid in it being 69%.
Ans. Mass percent of nitric acid in the sample = 69 % [Given]
Thus, 100 g of nitric acid contains 69 g of nitric acid by mass.
Molar mass of nitric acid (HNO3 )
= {1 + 14 + 3(16)} g 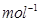
= 1 + 14 + 48
= 63 g 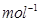
∴ Number of moles in 69 g of HNO3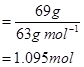
Volume of 100g of nitric acid solution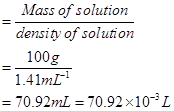
Concentration of nitric acid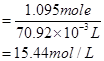
∴Concentration of nitric acid = 15.44 mol/L
7. How much copper can be obtained from 100 g of copper sulphate (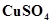 )?
)?
Ans. 1 mole of CuSO4 contains 1 mole of copper.
Molar mass of CuSO4 = (63.5) + (32.00) + 4(16.00)
= 63.5 + 32.00 + 64.00
= 159.5 g
159.5 g of CuSO4 contains 63.5 g of copper.
⇒ 100 g of CuSO4 will contain 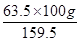 of copper.
of copper.
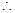 Amount of copper that can be obtained from 100 g CuSO4
Amount of copper that can be obtained from 100 g CuSO4 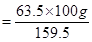
= 39.81 g
8. Determine the molecular formula of an oxide of iron in which the mass per cent of iron and oxygen are 69.9 and 30.1 respectively. Given that the molar mass of the oxide is 159.69 g 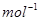 .
.
Ans. Mass percent of iron (Fe) = 69.9% (Given)
Mass percent of oxygen (O) = 30.1% (Given)
Number of moles of iron present in the oxide 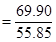
= 1.25
Number of moles of oxygen present in the oxide 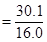
= 1.88
Ratio of iron to oxygen in the oxide,
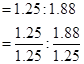
= 1: 1.5
= 2: 3
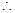 The empirical formula of the oxide is
The empirical formula of the oxide is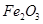 .
.
Empirical formula mass of 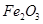 = [2(55.85) + 3(16.00)] g
= [2(55.85) + 3(16.00)] g
Molar mass of 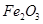 = 159.69 g
= 159.69 g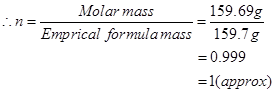
Molecular formula of a compound is obtained by multiplying the empirical formula with n.
Thus, the empirical formula of the given oxide is 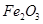 and n is 1.
and n is 1.
Hence, the molecular formula of the oxide is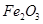 .
.
9. Calculate the atomic mass (average) of chlorine using the following data:

Ans. The average atomic mass of chlorine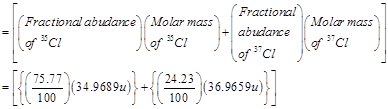
= 26.4959 + 8.9568
= 35.4527 u The average atomic mass of chlorine = 35.4527 u
The average atomic mass of chlorine = 35.4527 u
10. In three moles of ethane 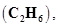 calculate the following:
calculate the following:
(i) Number of moles of carbon atoms.
(ii) Number of moles of hydrogen atoms.
(iii) Number of molecules of ethane.
Ans. (i) 1 mole of 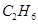 contains 2 moles of carbon atoms.
contains 2 moles of carbon atoms.
Number of moles of carbon atoms in 3 moles of 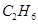
= 2 × 3 = 6
(ii) 1 mole of 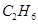 contains 6 moles of hydrogen atoms.
contains 6 moles of hydrogen atoms. Number of moles of carbon atoms in 3 moles of
Number of moles of carbon atoms in 3 moles of 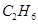
= 3 × 6 = 18
(iii) 1 mole of 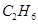 contains
contains 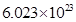 molecules of ethane.
molecules of ethane.
Number of molecules in 3 moles of 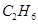
= 3 × 6.023 × 1023 = 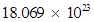
11. What is the concentration of sugar (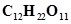 ) in mol
) in mol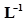 if its 20 g are dissolved in enough water to make a final volume up to 2 L?
if its 20 g are dissolved in enough water to make a final volume up to 2 L?
Ans. Molarity (M) of a solution is given by,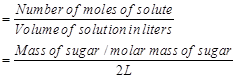
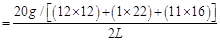
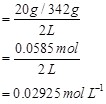
Molar concentration of sugar = 0.02925 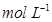
12. If the density of methanol is 0.793 kg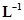 , what is its volume needed for making 2.5 L of its 0.25 M solution?
, what is its volume needed for making 2.5 L of its 0.25 M solution?
Ans. Molar mass of methanol (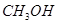 ) = (1 × 12) + (4 × 1) + (1 × 16)
) = (1 × 12) + (4 × 1) + (1 × 16)
= 32 g 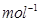
= 0.032 kg 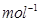
Molarity of methanol solution 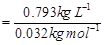
= 24.78 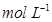
(Since density is mass per unit volume)
Applying, 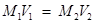
(Given solution) (Solution to be prepared)
(24.78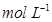 )
) 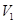 = (2.5 L) (0.25
= (2.5 L) (0.25 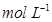 )
)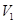 = 0.0252 L
= 0.0252 L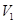 = 25.22 mL
= 25.22 mL
13. Pressure is determined as force per unit area of the surface. The SI unit of pressure, Pascal is as shown below: 1Pa = 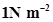 If mass of air at sea level is 1034 g
If mass of air at sea level is 1034 g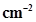 , calculate the pressure in Pascal.
, calculate the pressure in Pascal.
Ans. Pressure is defined as force acting per unit area of the surface.
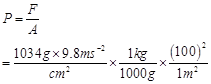
= 1.01332 × 105 kg 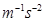
We know, 1 N = 1 kg
Then,
1 Pa = 1 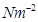 = 1 kg
= 1 kg 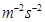
1 Pa = 1 kg 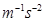
 Pressure =
Pressure = 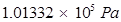
14. What is the SI unit of mass? How is it defined?
Ans. The SI unit of mass is kilogram (kg). 1 Kilogram is defined as the mass equal to the mass of the international prototype of kilogram.
15. Match the following prefixes with their multiples: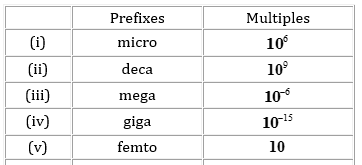
Ans.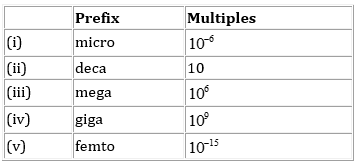
16. What do you mean by significant figures?
Ans. Significant figures are those meaningful digits that are known with certainty. They indicate uncertainty in an experiment or calculated value. For example, if 15.6 mL is the result of an experiment, then 15 is certain while 6 is uncertain, and the total number of significant figures are 3.
Hence, significant figures are defined as the total number of digits in a number including the last digit that represents the uncertainty of the result.
17. A sample of drinking water was found to be severely contaminated with chloroform, 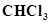 , supposed to be carcinogenic in nature. The level of contamination was 15 ppm (by mass).
, supposed to be carcinogenic in nature. The level of contamination was 15 ppm (by mass).
(i) Express this in percent by mass.
(ii) Determine the molality of chloroform in the water sample.
Ans. (i) 1 ppm is equivalent to 1 part out of 1 million (106) parts.
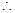 Mass percent of 15 ppm chloroform in water
Mass percent of 15 ppm chloroform in water
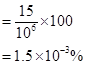
(ii) 100 g of the sample contains 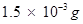 of
of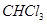 .
.
⇒ 1000 g of the sample contains 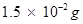 of
of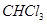 .
.
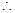 Molality of chloroform in water
Molality of chloroform in water
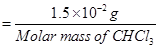
Molar mass of 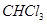 = 12.00 + 1.00 + 3(35.5)
= 12.00 + 1.00 + 3(35.5)
= 119.5 g 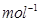
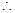 Molality of chloroform in water = 0.0125 ×
Molality of chloroform in water = 0.0125 × 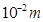
= 1.25 × 10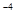 m
m
18. Express the following in the scientific notation:
(i) 0.0048
(ii) 234,000
(iii) 8008
(iv) 500.0
(v) 6.0012
Ans. (i) 0.0048 = 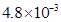
(ii) 234, 000 = 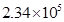
(iii) 8008 = 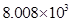
(iv) 500.0 = 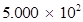
(v) 6.0012 = 6.0012
19. How many significant figures are present in the following?
(i) 0.0025
(ii) 208
(iii) 5005
(iv) 126,000
(v) 500.0
(vi) 2.0034
Ans. (i) 0.0025
There are 2 significant figures.
(ii) 208
There are 3 significant figures.
(iii) 5005
There are 4 significant figures.
(iv) 126,000
There are 3 significant figures.
(v) 500.0
There are 4 significant figures.
(vi) 2.0034
There are 5 significant figures.
20. Round up the following upto three significant figures:
(i) 34.216
(ii) 10.4107
(iii) 0.04597
(iv) 2808
Ans. (i) 34.2
(ii) 10.4
(iii) 0.0460
(iv) 2810
21. The following data are obtained when dinitrogen and dioxygen react together to form different compounds:
s.no | mass of dinitrogen(N2 ) | mass of dinitrogen(N2 ) |
| (i) | 14 g | 16 g |
| (ii) | 14 g | 32 g |
| (iii) | 28 g | 32 g |
| (iv) | 28 g | 80g |
(a) Which law of chemical combination is obeyed by the above experimental data? Give its statement.
(b) Fill in the blanks in the following conversions:
(i) 1 km = ...................... mm = ...................... pm
(ii) 1 mg = ...................... kg = ...................... ng
(iii) 1 mL = ...................... L = ...................... 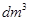
Ans. (a)If we fix the mass of dinitrogen at 28 g, then the masses of dioxygen that will combine with the fixed mass of dinitrogen are 32 g, 64 g, 32 g, and 80 g.
The masses of dioxygen bear a whole number ratio of 1:2:2:5. Hence, the given experimental data obeys the law of multiple proportions. The law states that if two elements combine to form more than one compound, then the masses of one element that combines with the fixed mass of another element are in the ratio of small whole numbers.
(b)(i) 1 km = 1 km ×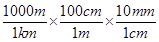
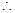 1 km = 106 mm
1 km = 106 mm
1 km = 1 km ×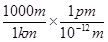
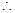 1 km = 1015 pm
1 km = 1015 pm
Hence, 1 km = 106 mm =1015 pm
(ii) 1 mg = 1 mg ×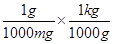
⇒ 1 mg = 10 kg
1 mg = 1 mg × 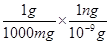
⇒ 1 mg = 106 ng
 1 mg =
1 mg = 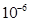 kg = 106 ng
kg = 106 ng
(iii) 1 mL = 1 mL × 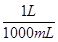
⇒ 1 mL = 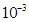 L
L
1 mL = 1 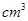 = 1
= 1 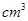
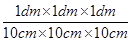
⇒ 1 mL 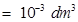
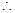 1 mL =
1 mL = 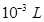 =
= 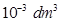
22. If the speed of light is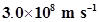 , calculate the distance covered by light in 2.00 ns.
, calculate the distance covered by light in 2.00 ns.
Ans. According to the question:
Time taken to cover the distance = 2.00 ns
Speed of light = 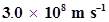
Distance travelled by light in 2.00 ns
= Speed of light x Time taken
= 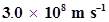
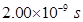
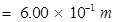 = 0.600 m
= 0.600 m
23. In a reaction A + 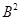 →
→ 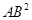
Identify the limiting reagent, if any, in the following reaction mixtures.
(i) 300 atoms of A + 200 molecules of B
(ii) 2 mol A + 3 mol B
(iii) 100 atoms of A + 100 molecules of B
(iv) 5 mol A + 2.5 mol B
(v) 2.5 mol A + 5 mol B
Ans. A limiting reagent determines the extent of a reaction. It is the reactant which is the first to get consumed during a reaction, thereby causing the reaction to stop and limiting the amount of products formed.
(i) According to the given reaction, 1 atom of A reacts with 1 molecule of B. Thus, 200 molecules of B will react with 200 atoms of A, thereby leaving 100 atoms of A unused. Hence, B is the limiting reagent.
(ii) According to the reaction, 1 mol of A reacts with 1 mol of B. Thus, 2 mol of A will react with only 2 mol of B. As a result, 1 mol of B will not be consumed. Hence, A is the limiting reagent.
(iii) According to the given reaction, 1 atom of A combines with 1 molecule of B. Thus, all 100 atoms of A will combine with all 100 molecules of B. Hence, the mixture is stoichiometric where no limiting reagent is present.
(iv) 1 mol of atom A combines with 1 mol of molecule B. Thus, 2.5 mol of B will combine with only 2.5 mol of A. As a result, 2.5 mol of A will be left as such. Hence, B is the limiting reagent.
(v) According to the reaction, 1 mol of atom A combines with 1 mol of molecule B. Thus, 2.5 mol of A will combine with only 2.5 mol of B and the remaining 2.5 mol of B will be left as such. Hence, A is the limiting reagent.
24. Dinitrogen and dihydrogen react with each other to produce ammonia according to the following chemical equation:
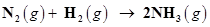
(i) Calculate the mass of ammonia produced if 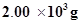 dinitrogen reacts with 1.00
dinitrogen reacts with 1.00 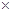 103 g of dihydrogen.
103 g of dihydrogen.
(ii) Will any of the two reactants remain unreacted?
(iii) If yes, which one and what would be its mass?
Ans. (i) Balancing the given chemical equation,
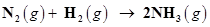
From the equation, 1 mole (28 g) of dinitrogen reacts with 3 mole (6 g) of dihydrogen to give 2 mole (34 g) of ammonia.
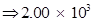 g of dinitrogen will react with
g of dinitrogen will react with 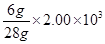 dihydrogen i.e.,
dihydrogen i.e.,
2.00 × 103 g of dinitrogen will react with 428.6 g of dihydrogen.
Given,
Amount of dihydrogen = 1.00 × 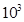 g
g
Hence, N2 is the limiting reagent.
 28 g of N2 produces 34 g of NH3
28 g of N2 produces 34 g of NH3
Hence, mass of ammonia produced by 2000 g of N2
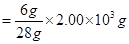
= 2428.57 g
(ii) N2 is the limiting reagent and H2 is the excess reagent. Hence, H2 will remain unreacted.
(iii) Mass of dihydrogen left unreacted 1.00* 103 g _ 428.6 g = 571.4 g
25. How are 0.50 mol Na2CO3 and 0.50 M Na2CO3 different?
Ans. Molar mass of Na2CO3 = (2 × 23) + 12.00 + (3× 16)
= 106 g 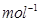
Now, 1 mole of Na2CO3 means 106 g of Na2CO3.
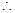 0.5 mol of Na2CO3
0.5 mol of Na2CO3
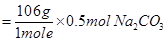
= 53 g of Na2CO3
⇒ 0.50 M of Na2CO3
= 0.50 mol/L of Na2CO3
Hence, 0.50 mol of Na2CO3 is present in 1 L of water or 53 g of Na2CO3 is present in 1 L of water.
