Minerals and Rocks - Revision Notes
Class 11 Geography
Chapter-5 Minerals and Rocks
This unit deals with : Minerals, elements, characteristics of minerals such as crystal form cleavage, fracture, lustre, important minerals such as feldspar, quartz, pyroxene, colour, streak, transparency, structure, hardness specific grvity, amphibole, mica, olivine and their characteristics classification of minerals, rocks, igneous, sedimentary, metamorphic rocks rock cycle Minerals found in the crust are in solid form where as in intrior they are in liquid form 98% of the crust consist of eight elements
- oxygen
- Aluminium
- Sodium
- Potassium
- Iron.
- Calcium
- Magnes the rest is constituted by titanium, hydrogen, phosphorous, manganese, sulphur carbon, nickel & other elements
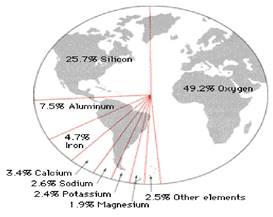
Table 5.1: The major Elements of the Earth’s Crust
| Sl. No. | Elements | By Weight(%) |
| 1. 2. 3. 4. 5. 6. 7. 8. 9. | Oxygen Silicon Aluminium Iron Calcium Sodium Potassium Magnesium Others | 46.60 27.72 8.13 5.00 3.36 2.83 2.59 2.09 1.4 |
Minerals: The elements in the earth’s crust are rarely found exclusively but are usually combined with other elements to make various substances. These substances are recognised as minerals. Minerals are substances that are formed naturally in the Earth. Minerals are usually solid, have a crystal structure, inorganic and form naturally by geological processes. The study of minerals is called mineralogy. A mineral can be made of single chemical element or more usually a compound. Naturally occuring inorganic substance having an orderly atomic structure and a definite chemical composition and physical properties.
It is composed of two or three minerals /single element ex. S,Cu, Ag,Au, Graphite.
There are at least 2000 minerals in the crust. There are at least 2,000 minerals that have been named and identified in the earth crust
The basic source of all minerals is the hot magma in the interior of the earth. Coal, petroleum and natural gas are organic minerals
PHYSICAL CHARACTERISTICS OF MINERALS
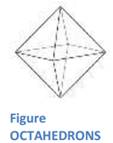
I. EXTERNAL CRYSTAL FORM: The crystal form of a mineral is a set of faces that have a definite geometric relationship to one another. What most people call a "crystal" is a mineral with well-developed crystal faces. If minerals were always able to develop their characteris-tic crystal forms, mineral identification would be a much sim-pler task. In rocks, however, most minerals grow while competing for space with other minerals. lnternal arrangement of molecules-cube, octahedrons, hexagonal, prisms.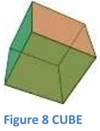

II. CLEAVAGE: After minerals are formed, they have a tendency to split or break along definite planes of weakness. This property is called cleavage. These planes of weakness are closely related to the internal structure of the mineral, and are usually, but not always, parallel to crystal faces or possible crystal faces. Minerals may have one, two, three, four, or six directions of cleavage. These cleavage forms are (1) cubic, (2) octahedral, (3) dodecahedral, (4) rhombohedral, (5) prismatic, and (6) pinacoidal. Minerals that break easily along these lines of weakness yield shiny surfaces. Many crystals do not cleave, but fracture or break instead. Quartz, for example, forms well-developed crystal faces but does not cleave at all; instead it fractures or breaks randomly with a conchoidal fracture.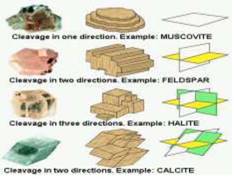
FRACTURE: Conchoidal fracture
Obsidian
Conchoidal fracture : It is a curved breakage that resembles the concentric ripples of a mussel shell. It often occurs in amorphous or fine-grained minerals such as flint, opal or obsidian, but may also occur in crystalline minerals such as quartz. Internal molecular arrangement so complex there are no planes of molecules; the crystal will break in an irregular manner, not along planes of cleavage
Earthy fracture
Limonite
Limonite is an iron ore consisting of a mixture of hydrated iron(III) oxide-hydroxides in varying composition. Earthy fracture is reminiscent of freshly broken soU. It is frequently seen in relatively soft, loosely bound minerals, such as limonite, kaolinite and aluminite.
Hackly fracture
Native copper
Native copper is an uncombined form of copper which occurs as a natural mineral. Copper is one of the few metallic elements to occur in native form, although it most commonly occurs in oxidized states and mixed with other elements. Hackly fracture (also known as jagged fracture) is jagged, sharp and uneven. It occurs when metals are torn, and so is often encountered in native metals such as copper and silver.
Splintery fracture
Chrysotile
Chrysotile or white asbestos is the most commonly encountered form of asbestos, accounting for approximately 95% of the asbestos in the United States and a similar proportion in other countries. Splintery fracture comprises sharp elongated points. It is particularly seen in fibrous minerals such as chrysotile, but may also occur in nonfibrous minerals such as kyanite.
Uneven fracture

Magnetite
Magnetite is a mineral and one of the main iron ores. With the chemical formula Fe₃O₄, it is one of the oxides of iron. Magnetite is ferrimagnetic; it is attracted to a magnet and can be magnetized to become a permanent magnet itself. Uneven fracture is self descriptive. It occurs in a wide range of minerals including arsenopyrite, pyrite and magnetite. The crystal breaks inan irregular manner not along the planes
LUSTURE: Lustre or luster is the way light interacts with the surface of a crystal, rock, or mineral. The word traces its origins back to the latin lux, meaning "light", and generally implies radiance, gloss, or brilliance. A range of terms are used to describe lustre, such as earthy, metallic, greasy, and silky. Luster refers to the brightness of light reflected from the mineral's surface. The main types of luster are metallic and nonmetallic. Some of the more important nonmetallic lusters are:
- Adamantine: brilliant, like that of a diamond.
- Earthy: dull, like kaolin.
- Silky: having the sheen of silk, like satin spar, a variety of gypsum.
- Greasy: oily appearance.
- Resinous: waxy appearance, like sphalerite.
- Vitreous: the appearance of broken glass, like quartz.
- Nacreous (pearly): like mother of pearl; for example, pearly luster on fossil gastropods and cephalopods.
Adamantine lustre
Cut diamonds
Adamantine minerals possess a superlative lustre, which is most notably seen in diamond. Such minerals are transparent or translucent, and have a high refractive index (of 1.9 or more)Minerals with a true adamantine lustre are uncommon, with examples being cerussite and zircon.
Minerals with a lesser (but still relatively high) degree of luster are referred to as subadamantine, with some examples being garnet and corundum Dull lustre
Kaolinite
Dull (or earthy) minerals exhibit little to no luster, due to coarse granulations which scatter light in all directions, approximating a Lambertian reflector. An example is kaolinite. A distinction is sometimes drawn between dull minerals and earthy minerals, with the latter being coarser, and having even less lustre.
Greasy lustre
Moss opal
Moss opal is a milky white to brownish opal with dark greenish inclusions that resemble moss. Moss opal may also be referred to as "dendritic opal" because of the tree-like appearance of the inclusions, or "dendrites", which comes from the Greek word, "dendron", meaning "tree". Greasy minerals resemble fat or grease. A greasy lustre often occurs in minerals containing a great abundance of microscopic inclusions, with examples including opal and cordierite. Many minerals with a greasy lustre also feel greasy to the touch.
Metallic lustre
Pyrite
The mineral pyrite, or iron pyrite, also known as fool's gold, is an iron sulfide with the chemical formula FeS2. This mineral's metallic luster and pale brass-yellow hue give it a superficial resemblance to gold, hence the well-known nickname of fool's gold. Metallic (or splendant) minerals have the lustre of polished metal, and with ideal surfaces will work as a reflective surface. Examples include Salena Pyrite and magnetite.
Pearly lustre
Muscovite
Muscovite (also known as common mica, is in glass, or potash mica) is a hydrated phyllosilicate mineral of aluminium and potassium with formula KAl2(AlSi3O10)(F,OH)2, or (KF)2(Al2O3)3(SiO2)6(H2O). It has a highly perfect basal cleavage yielding remarkably thin laminae (sheets) which are often highly elastic. Pearly minerals consist of thin transparent co-planar sheets. Light reflecting from these layers give them a lustre reminiscent of pearls. Such minerals possess perfect cleavage, with examples including muscovite and stilbite.
Resinous Lustre
Amber
Amber is fossilized tree resin, which has been appreciated for its color and natural beauty since Neolithic times. Much valued from antiquity to the present as a gemstone, amber is made into a variety of decorative objects. Amber is used in jewellry. Resinous minerals have the appearance of resin, chewing gum or (smooth surfaced) plastic. A principal example is amber, which is a form of fossilized resin.
Silky lustre
Satin spar variety of gypsum
Silky minerals have a parallel arrangement of extremely fine fibres, giving them a lustre reminiscent of sUk. Examples include asbestos, ulexite and the satin spar variety of gypsum. A fibrous lustre is similar, but has a coarser texture.
Submetallic lustre
Sphalerite Submetallic minerals have similar lustre to metal, but are duller and less reflective. A submetallic lustre often occurs in nearopaque minerals with very high refractive indices,[9] such as sphalerite, cinnabar and cuprite.
Vitreous lustre
Quartz
Quartz is a chemical compound consisting of one part silicon and two parts oxygen. It is silicon dioxide (SiO2). It is the most abundant mineral found at Earth's surface, and its unique properties make it one of the most useful natural substances. Rock crystal quartz: Transparent "rock crystal" quartz. Vitreous minerals have the lustre of glass. (The term is derived from the Latin for glass, vitrum.) This type of lustre is one of the most commonly seen, and occurs in transparent or translucent minerals with relatively low refractive indices. Common examples include calcite, quartz, topaz, beryl, tourmaline and fluorite, among others.
Waxy lustre
Jade
Jade is an ornamental rock, mostly known for its green varieties, which feature prominently in ancient Asian art. Waxy minerals have a lustre resembling wax. Examples include iade and chalcedony.
Optical phenomena Asterism
Sapphire cabochon
Asterism or star stone, is a name applied to the phenomenon of gemstones ... An asterism is an optical phenomenon displayed by some rubies, sapphires, and other gems. Asterism is the display of a star-shaped luminous area. It is seen in some sapphires and rubies, where it is caused by impurities of rutile. It can also occur in garnet, diopside and spinel.
Aventurescence
Aventurine is a form of quartz, characterised by its translucency and the presence of platy mineral inclusions that give a shimmering or glistening effect termed aventurescence. Aventurine Aventurescence (or aventurization) is a reflectance effect like that of glitter. It arises from minute, preferentially oriented mineral platelets within the material. These platelets are so numerous that they also influence the material's body colour. In aventurine quartz, chrome-bearing fuchsite makes for a green stone and various iron oxides make for a red stone.
Chatoyancy
Tiger's eye
The term "cat's eye", when used by itself as the name of a gemstone, refers to a cat's eye chrysoberyl. It is also used as an adjective which indicates the chatoyance phenomenon in another stone, e.g., cat's eye aquamarine. Chatoyant minerals display luminous bands, which appear to move as the specimen is rotated. Such minerals are composed of parallel fibers (or contain fibrous voids or inclusions), which reflect light into a direction perpendicular to their orientation, thus forming narrow bands of light. The most famous examples are tiger's eye and cymophane, but the effect may also occur in other minerals such as aquamarine, moonstone and tourmaline.
Colour change
Alexandrite
Color change is most commonly found in Alexandrite, a variety of chrysoberyl gemstones. Other gems also occur in color-change varieties, including (but not limited to) sapphire, garnet, spinel. Alexandrite displays a color change dependent upon light, along with strong pleochroism. The gem results from small scale replacement of aluminum by chromium oxide, which is responsible for alexandrite's characteristic green to red color change. Alexandrite from the Ural Mountains in Russia is green by daylight and red by incandescent light. Other varieties of alexandrite may be yellowish or pink in daylight and a columbine or raspberry red by incandescent light. The optimum or "ideal" color change would be fine emerald green to fine purplish red, but this is exceedingly rare. Schiller Labradorite
Schiller, from German for "twinkle", is a term used to describe the metallic iridescence originating from below the surface of a stone, that occurs when light is reflected between layers of minerals. It is seen in moonstone and labradorite and is very similar to adularescence and aventurescence.
appearance of material without regard to clolour-metallic silky glossy
COLOUR: Some minerals have characteristic colour determined by their molecular structure — malachite, azurite, chalcopyrite etc., and some minerals are coloured by impurities. For example, because of impurities quartz may be white, green, red, yellow etc. 
STREAK: Colour of the ground powder of any mineral. It may be of the same colour as the mineral or may differ — malachite is green and gives green streak, fluorite is purple or green but gives a white streak.
TRANSPARENCY: Transparent: light rays pass through so that objects can be seen plainly; translucent — light rays pass through but will get diffused so that objects cannot be seen; opaque — light will not pass at all.
Terminology: Opaque - no light can pass through the mineral;
Translucent - light can pass through the mineral but is diffused so that images cannot be seen clearly;
Transparent- light can pass through the mineral and images can be seen clearly.


- Transparency -light rays pass through
- translucent-light pass through but diffused
- opaque- light doesnot pass through.
Magnetite
III. STRUCTURE: Particular arrangement of the individual crystals; fine, medium or coarse grained; fibrous — separable, divergent, radiating.
HARDNESS: as corundum. The table below shows comparison with absolute hardness measured by a sclerometer, with pictorial examples.
| Mohs hardness | Mineral | Chemical formula | Absolute hardness | image |
| 1 | Talc | mg3Si4O10(OH)2 | 1 |  |
| 2 | Gypsum | CaSo42H2O | 3 |  |
| 3 | Calcite | CaCO3 | 9 |  |
| 4 | Fluorite | CaF2 | 21 |  |
| 5 | Apatite | Ca(PO4)3(OH+Cl-F+) | 48 |  |
| 6 | Orthoclase Feldspar | KAlSi3O3 | 72 |  |
| 7 | Quartz | SiO2 | 100 |  |
| 8 | Topaz | Al2SiO4(OH-,F-)2 | 200 |  |
| 9 | Corundum | Al2O3 | 400 |  |
| 10 | diamond | C | 1600 |  |
IV. Relative resistence being scratched ten minerals are selected to measure the degree of hardness from 1to 10
- Talc
- gypsum
- calcite
- fluorite
- apatite
- feldspar
- quartz
- topaz
- corundum
- diamond. ex. fingre nail has 2.5 hardness knife has 5.5 hardness

SPECIFIC GRAVITY: The ratio between the weight of a given object and the weight of an equal volume of water ; object weighed in air and then weighed in water and divide weight in an airby the difference of the two weights.
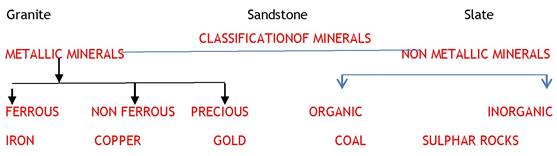
IMPORTANT MINERALS OF THE EARTH CRUST
FELDSPAR: Feldspar is the name given to a group of minerals distinguished by the presence of alumina and silica (SiO2) in their chemistry. Half of the earth’s crust is composed of feldspar. It has light cream to salmon pink colour. It is used in ceramics and glass making.
QUARTZ: Quartz is a chemical compound consisting of one part silicon and two parts oxygen. It is silicon dioxide (SiO2). It is the most abundant mineral found at Earth's surface, and its unique properties make it one of the most useful natural substances. Rock crystal quartz: Transparent "rock crystal" quartz. Important component of sand granite.consists of silica, hard mineral, insoluble in water it is white ore coloruless used INRADO OR RADOR
PROXENE: The pyroxenes most commonly occur in intermediate to ultra-mafic igneous rocks, although they are also common in some medium-grade to high-grade metamorphic rocks. Pyroxene consists of calcium, aluminum, magnesium, iron and silica. Pyroxene forms 10 per cent of the earth’s crust. It is commonly found in meteorites. It is in green or black colour.
OLIVINE: Olivine is one of the most common minerals in the earth, and is a major rock forming mineral. Despite this, good specimens and large crystals are uncommon and sought after. Magnesium, iron and silica are major elements of olivine. It is used in jewellery. It is usually a greenish crystal, often found in basaltic rocks. 
AMPHIBOLE: Amphibole is the name of an important group of generally dark-colored, inosilicate minerals, forming prism or needlelike crystals, composed of double chain SiO. Aluminium, calcium, silica, iron, magnesium are the major elements of amphiboles. They form 7 per cent of the earth’s crust. It is in green or black colour and is used in asbestos industry. Hornblende is another form of amphiboles.
MICA: It comprises of potassium, aluminium, magnesium, iron, silica etc. It forms 4 per cent of the earth’s crust. It is commonly found in igneous and metamorphic rocks. It is used in electrical instruments.
IGNEOUS ROCKS : Igneous rock is formed when magma cools and solidifies, it may do this above or below the Earth's surface. Magma can be forced into rocks, blown out in volcanic explosions or forced to the surface as lava. The atoms and molecules of melted minerals are what make up magma.
- They are primary rocks.
- formed due to cooling of lava
- They are two types intrusive & extrusive rocks. Extrusive rocks have small grains because of sudden cooling intrusive rocks have bigger grains due to slow cooling.
- No layers
- Do not contain fossils.
- They are hard.
- Do not allow water to percolate through them.
SEDIMENTARY ROCKS: The word ‘sedimentary’ is derived from the Latin word sedimentum, which means settling. Rocks (igneous, sedimentary and metamorphic) of the earth’s surface are exposed to denudational agents, and are broken up into various sizes of fragments. In many sedimentary rocks, the layers of deposits retain their characteristics even after lithification.
- Formed Due To Sedimentation
- Consists Of Layers
- Contain Fossils
- The Process Of Sedimentary Rock Formation Is Called Lithification
- They Are Three Types.
- Mechanically Formed
- Chemically Formed
- Organically Formed.
METAMORPHIC ROCKS: A metamorphic rock is a result of a transformation of a pre-existing rock. The original rock is subjected to very high heat and pressure, which cause obvious physical and/or chemical changes. Examples of these rock types include marble, slate, gneiss, schist.
- Formed Due To Recrystalization.
- Formed Due To Pressure And Temperature.
- Very Smooth.
- Consists of layers some times very precious stones.
Type of rock
Igneous rock Sedimentary rockMetamorphic rock I


Rocks are aggregate of one or more minerals, they may be hard or soft in varied colours, they do not have definite chemical composition.
Petrology is the science of rocks. Petrologist who studies the scientific methods of rocks.
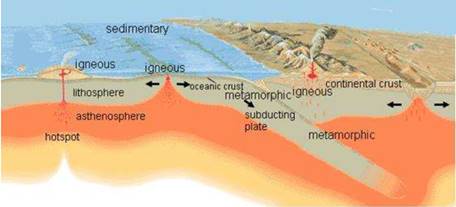
ROCK CYCLE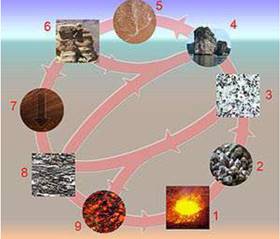
A diagram of the rock cycle. Legend:
- magma;
- crystallization (freezing of rock);
- igneous rocks;
- erosion;
- sedimentation;
- sediments & sedimentary rocks;
- tectonic burial and metamorphism;
- metamorphic rocks;
- melting. magma;
Rock cycle is a continuous process through which old rocks are converted into new rocks. Igneous rocks are changed into metamorphicor sedimentary rocks. Metamorphic rocks afurther change into magma.
