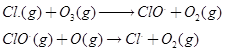Environmental Chemistry - Test Papers
CBSE TEST PAPER 01
CLASS XI CHEMISTRY (Environmental Chemistry)
General Instruction:
- All questions are compulsory.
- Marks are given alongwith their questions.
1. What is troposphere? [1]
2. What is the role of ozone layer in the stratosphere? [2]
3. Name some gaseous air pollutants. [1]
4. What are the diseases caused by sulphur dioxide? [1]
5. What includes stratospheric pollutants? Give examples. [2]
6. What are the harmful effects of oxides of nitrogen in atmosphere? [3]
7. Why is carbon monoxide considered to be poisonous? [2]
8. What are the ill-effects of hydrocarbons? [2]
9. What are the reactions involved for ozone layer depletion in the atmosphere? [3]
CBSE TEST PAPER 01
CLASS XI CHEMISTRY (Environmental Chemistry)
[ANSWERS]
Ans 01. The lowest region atmosphere in which the human beings along with other organisms live is called troposphere. It extends upto the height of 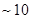 km from sea level.
km from sea level.
Ans 02. The presence of ozone in the stratosphere prevents about 99.5% of the sun’s harmful ultraviolet (uv) radiations from reaching the earth’s surface and thereby protecting humans and other animals from its effect.
Ans 03. Gaseous air pollutants are oxides of sulphur, nitrogen and carbon, hydrogen sulphide, hydrocarbons, ozone and other oxidants.
Ans 04. Sulphur dioxide causes respiratory diseases eg. asthma, bronchitis, emphysema in human beings, sulphur dioxide causes irritation to the eyes, resulting in tears and redness.
Ans 05. Depletion of ozone layer in stratospheres leading to reach harmful uv radiation on earth is the result of stratospheric pollution. The presence of chloro fluoro carbon compounds in the atmosphere is responsible for this depletion.
Ans 06. (i) High concentration of NO2 in atmosphere is harmful to plants resulting in leaf spotting, retardation of photosynthetic activity and also suppression the vegetation growth.
(ii) Nitrogen dioxide (NO2) results in respiratory problems in human beings and leads to bronchitis. It causes acid rain. Produce photochemical smog.
(iii) Oxides of nitrogen have harmful effects on the nylon, rayon and cotton yarns and also cause cracks in rubber.
(iv)They also react with react with ozone (O3) present in the atmosphere, and, their decrease the density of ozone.
Ans 07. Carbon monoxide binds to hemoglobin to form carboxyl – haemoglobin, which is about 300 times more stable than the oxygen – haemoglobin complex. In blood when the concentration of carboxyl hemoglobin is greatly reduced. This oxygen deficiency, results into headache, weak eyesight, nervousness and cardiovascular disorder.
Ans 08. Hydrocarbons are carcinogenic i. e; they cause cancer. They harm plants by causing ageing, breakdown of tissues and shedding of leaves flowers and trigs.
Ans 09.